https://www.wateronline.com/doc/capitalizing-on-evolving-pfas-removal-options-0001
Granular activated carbon (GAC) and ion exchange (IX) resin have long histories in removing a variety of per- and polyfluoroalkyl substances (PFAS), known as ‘forever chemicals.’ But choosing optimal treatment vessel and media efficiency depends on the specifics of the application. Here are some performance considerations of evolving options that can pay dividends for first-time and experienced PFAS removal operators alike.

Build On The Basics
For years, GAC and IX resin have been used individually or in combination for their specific attributes in removing various PFAS compounds — e.g., perfluorooctanoic acid (PFOA), perfluorooctane sulfonate (PFOS), etc. — and other source water components. Because these technologies are not one-size-fits-all solutions, however, first-time users should familiarize themselves with core treatment media and equipment attributes before making their selection. And because the mechanics of different system designs can impact operating costs and system longevity, even experienced users should reevaluate the nuances of corrosion resistance and pressure loss before upgrading capacity or replacing older systems (Figure 1).
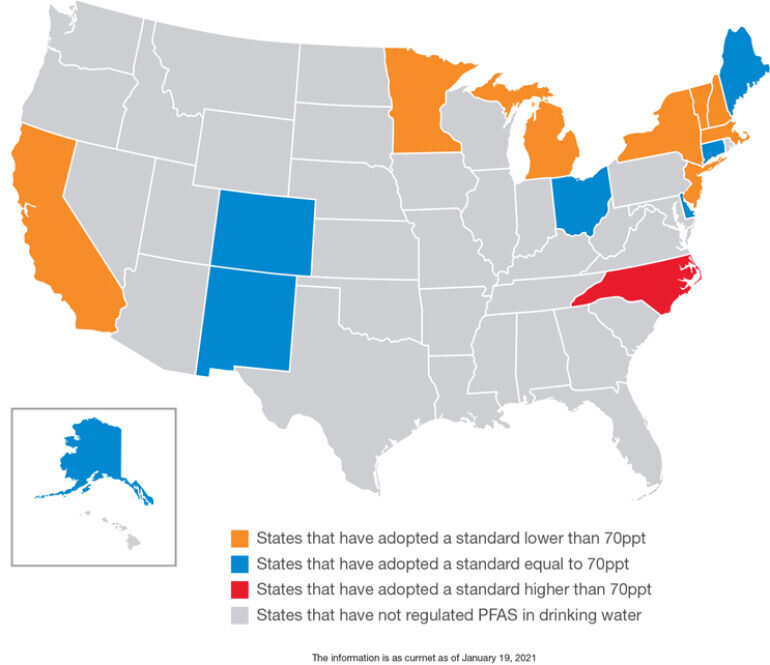
Figure 1. As shown in the map above, changing regulatory standards in certain states are generating increased pressures to lower PFAS exposure in drinking water. As that trend expands, smaller water treatment plants (WTPs) and more operators will need to identify and implement more effective PFAS removal technologies.
Match The Best Media Options To The Water Conditions
Choosing an optimum PFAS removal design should account for the overall composition of the source water, the type and concentration of PFAS compounds it contains, and critical media performance attributes relative to those specific conditions. Making the most cost-effective choice also requires considering the physics of the treatment equipment and the impact on energy efficiency. Start by comparing these critical performance attributes:
- Long-Chain/Short-Chain Molecules. The chemical composition of targeted contaminants is a primary consideration because of differences in how long-chain PFAS molecules (six or more carbon atoms in their structure) and short-chain molecules (five or fewer carbon atoms) react with carbon and resin media.
- GAC. While GAC has the capacity to adsorb both short-chain and long-chain PFAS molecules, it typically performs better at removing long-chain PFAS molecules quickly. It is also more effective in coping with heavier total organic carbon (TOC) and total suspended solids (TSS) loads in the water.
- IX Resins. In contrast to GAC adsorption, which acts only on the fluorinated tail of the molecule, IX resins involve both adsorption acting on the hydrophobic tail of the molecule and ion exchange acting on the ionized head of the molecule (Figure 2). This qualifies them to attract both short-chain and long-chain PFAS molecules quickly.
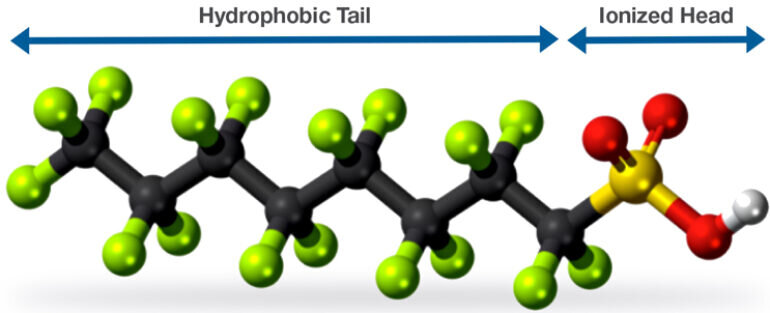
Figure 2. Even though both GAC and IX resin can capture both long- and short-chain PFAS molecules, efficient PFAS removal is a numbers game. Long-chain molecules easily adsorb to the carbon surface inside larger GAC pores, but short-chain molecules can pass through a GAC bed without adsorbing to the media because they are lighter and have weaker surface attraction. IX resins are well-suited to adsorb both short- and long-chain molecules with empty bed contact time (EBCT) requirements much shorter than those of GAC — on the order of 2 to 4 minutes vs. 10 to 15 minutes for GAC.
- Other Water Characteristics. The long/short-chain nature of the PFAS present requires media choices to be made in accordance with the total source water profile. For example, high TOC levels (> 2 ppm) can cause problems with IX resins that attract competing anions like sulfate, nitrate, phosphate, and silicates — thereby shortening the service life of the media for attracting targeted PFAS compounds.
- Media Cost Projections. Testing GAC performance through rapid, small-scale column tests (RSSCT) enables utilities to use laboratory-scale testing to collect two years’ worth of full-scale-plant GAC performance data in about two weeks. Doing so helps prospective users evaluate how various GAC media formats perform with their particular source water, project expected media life, and calculate long-term operating expenses (OPEX) for media replacement. Spiking the source water sample with a PFOA sample tagged at a specific radio frequency makes it easier to identify exactly when breakthrough occurs in the GAC column. By contrast, resin testing requires a longer (four-month) test at pilot plant scale. Either way, media selection should be evaluated on cost per unit of contaminant removed, not cost per unit weight of media.
- Single- Or Tandem-Treatment Installations. GAC and IX can each be applied as independent treatments, but in a fraction of cases where neither media format offers a total solution, the two can be used in sequence to handle extremes that neither technology alone can solve. In those situations, GAC treatment is typically used first as the workhorse to handle excess TOCs that would quickly overwhelm IX resins, and IX resins are used to polish the remaining short-chain PFAS compounds.
- Dealing With Moving Targets. As laboratory detection capabilities have improved, ‘non-detect’ levels that were 8 parts per trillion (ppt) just a few years ago are now down to 2 ppt and on their way to 0.8 ppt. This gives utilities trying to reach non-detect status new reasons to consider adding a supplemental IX system to help their existing 5- or 10-year-old GAC systems achieve higher levels of removal.
- Contaminant Disposal And Media Reuse. While not directly related to the PFAS removal process, the cost of handling spent media and destroying/disposing the captured PFAS can be important factors in media selection and overall lifecycle cost. Incineration is available for both media, but only GAC offers the advantage of cost-efficient reactivation.
Evaluate System Attributes Beyond Media, Too
Aside from the unique interaction of water composition and media being used to treat it, there are additional considerations related to energy-efficient performance and long-term OPEX implications. These include several areas where PFAS removal systems have evolved in recent years and where investments in new systems can pay dividends for years to come. Some of these options can make a strong case for evaluating equipment procurement separately from media procurement.
- Energy Concerns. Energy costs for pumping water are a big part of any water treatment plant (WTP) budget. Beyond evaluating PFAS removal efficiency, it also pays to evaluate the energy efficiency at which a system performs that task. Designs that reduce pressure drop through the system demand less energy to treat a given volume of water, thereby minimizing OPEX concerns over the life of the investment.
- System Construction. For operators concerned about satisfying NSF-61 requirements restricting the levels of lead contained in construction materials and the levels of lead that can leach out of the system, buying an NSF-61 certified system can put a decision-maker’s mind at ease (Figure 3).
- System Longevity. Corrosion is a valid concern for any system constantly exposed to flowing water 24/7 over a decades-long service life. Designs that use better corrosion-resistant construction materials or simplify access for epoxy-coating corrosion protection can pay dividends in terms of system longevity.

Figure 3. Unlike equipment described as “compliant with NSF-61,” complete PFAS removal systems that are fully NSF-61 certified eliminate the need for utilities to devote time and effort to documenting the lead-free performance of each component according to Safe Drinking Water Act (SDWA) standards.
Building A Better PFAS Strategy
Organizations facing their first PFAS removal system decision or changing regulations can benefit from consulting with experienced suppliers offering an extensive knowledge base and decades of diverse, hands-on environmental-remediation experience.


